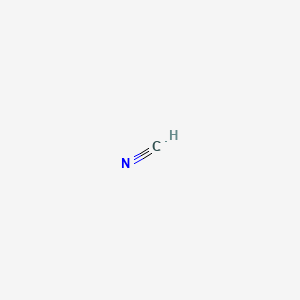
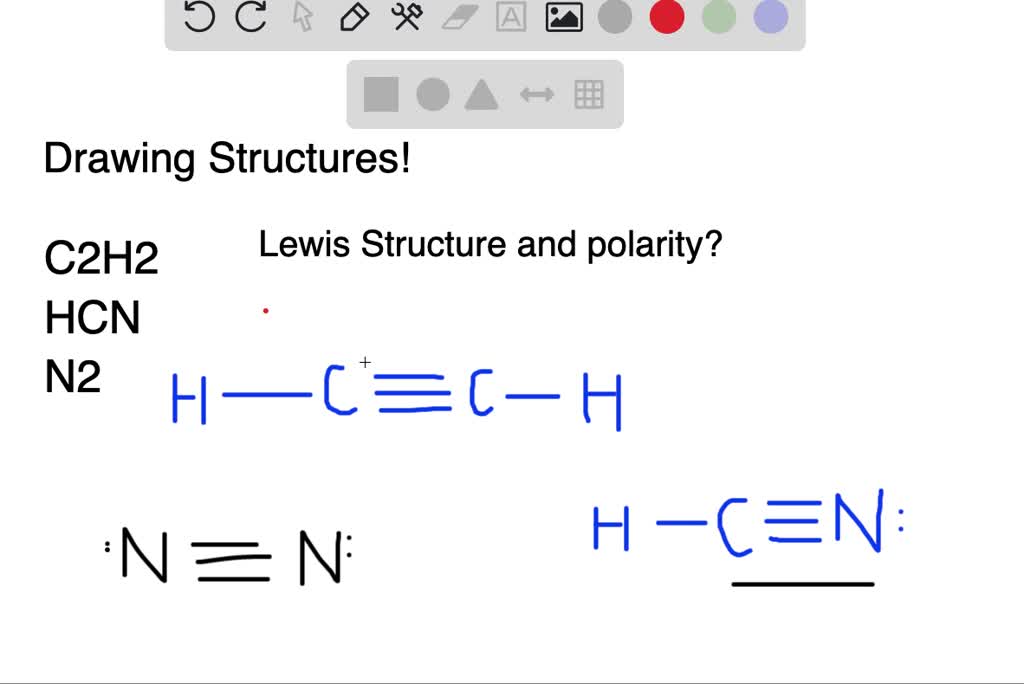
39 lewis diagram for hcn
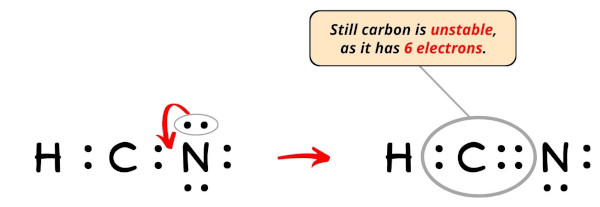
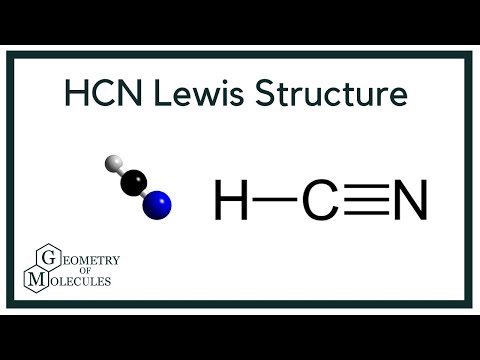
Chemical Bonding: HCN Lewis Structure - The Geoexchange We have a total of ten valence electrons for the HCN Lewis structure. We'll put two between atoms to form chemical bonds, so we've used four, then we'll go ...Oct 1, 2013 · Uploaded by Wayne Breslyn www2.chemistry.msu.edu › faculty › reuschorganic problems - Michigan State University 31 A Lewis formula for diazomethane, CH 2 N 2, is shown on the left below. Which of the formulas within the brackets would be considered a proper resonance contributor to this structure ? 32 The curved arrows in the formula on the left represent a chemical reaction.
quizlet.com › 477461106 › chemmy-flash-cardschemmy Flashcards - Quizlet The equilibrium constants (Ka) for HCN and HF in H2O at 25°C are 6.2 ´ 10-10 and 7.2 ´ 10-4, respectively. The relative order of base strengths is: CN- > F- > H2O
Lewis diagram for hcn
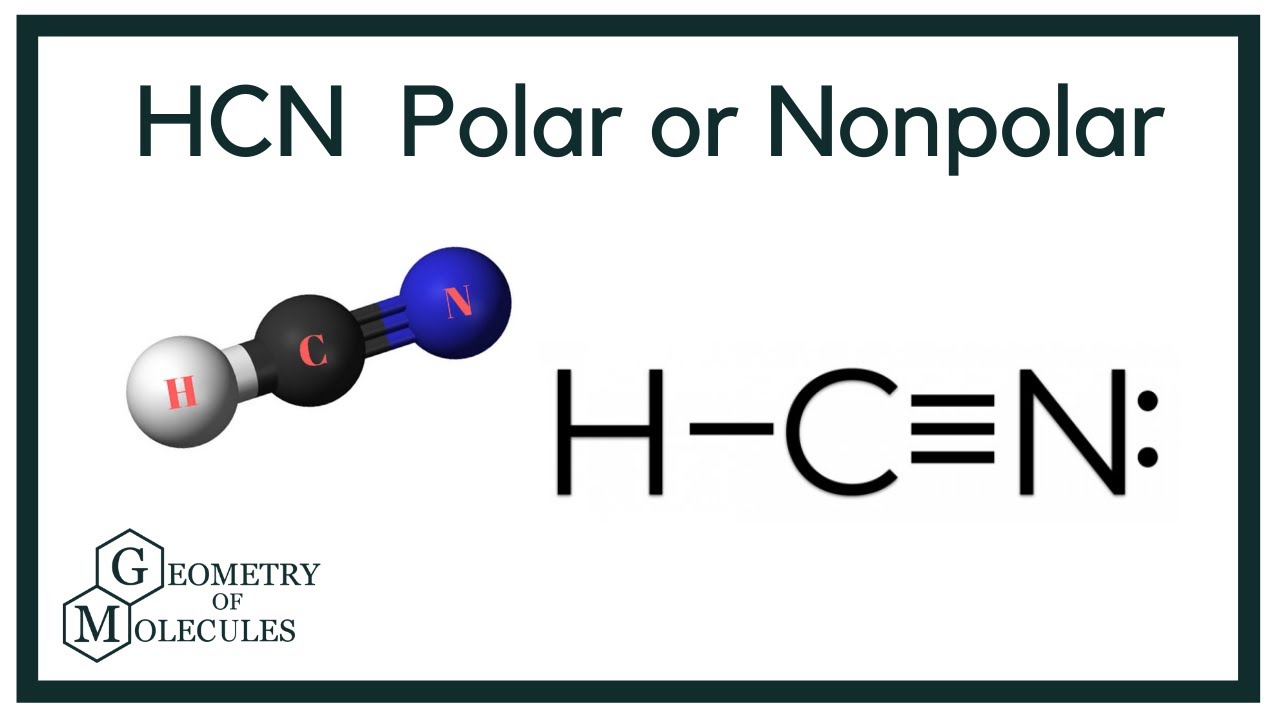
www2.chemistry.msu.edu › faculty › reuschAlkyl Halide Reactivity - Michigan State University An energy diagram for the single-step bimolecular E2 mechanism is shown on the right. We should be aware that the E2 transition state is less well defined than is that of S N 2 reactions. More bonds are being broken and formed, with the possibility of a continuum of states in which the extent of C–H and C–X bond-breaking and C=C bond-making ... HCN Lewis Structure, Molecular Geometry, Shape, and Polarity Carbon forms one single bond with the Hydrogen atom and forms a triple bond with the Nitrogen atom. · HCN has a total of 10 valence electrons.Jul 23, 2021 · Uploaded by Geometry of Molecules quizlet.com › 538030724 › module-two-chem-101Module Two Chem 101 Problems Flashcards - Quizlet A Lewis structure for CH₃CHCH₂ is shown below on the left. Predict whether bonding angles A and B will be equal to, greater than, or less than the ideal bonding angles according to the VSEPR model. (image 24) A. A-equal, B-equal B. A- greater than, B-less than C. A- less than, B- greater than D. A- less than, B- less than E.
Lewis diagram for hcn. socratic.org › questions › a-woman-at-an-airport-isA woman at an airport is towing her 20.0-kg ... - Socratic.org Jun 15, 2018 · b. theta = 55.2^@ c. "Normal force" = 167.3 N a. I will leave this task to you. My work on b and c should give you clues on what the free-body diagram needs to include. b. Since she is going at constant speed, none of her force is going to acceleration and none of the friction is making her decelerate. Therefore we can say that the horizontal component of her 35.0 N is equal to, but opposite ... jabchem.org.uk › chemistry › advancedhigher*X273/13/02* - JABchem The molecular orbital diagram for oxygen is shown. The anti-bonding orbitals are denoted by *. The bond order for a molecule of oxygen is A 0 B 1 C 2 D 3. O atomic orbitals O atomic orbitals O 2 molecular orbitals σ* σ* ɰ* ɰ σ σ 2p 2p 2s 2s Increasing Energy bond order = (number of bonding electrons – number of anti-bonding electrons) socratic.org › questions › by-writing-molecularBy writing molecular orbital configuration for NO,CO,O2 ... Mar 18, 2018 · The MO diagram of #"NO"# is: The last electron is in fact in the #2b_1# antibonding MO, so the bond order of #"NO"# has decreased by #1/2# relative to #"NO"^(+)# or #"CO"#. Therefore the bond order is #color(blue)(2.5)#. That corresponds to the Lewis structure of: #:dot"N"=ddot"O":# with a resonance hybrid structure of: quizlet.com › 538030724 › module-two-chem-101Module Two Chem 101 Problems Flashcards - Quizlet A Lewis structure for CH₃CHCH₂ is shown below on the left. Predict whether bonding angles A and B will be equal to, greater than, or less than the ideal bonding angles according to the VSEPR model. (image 24) A. A-equal, B-equal B. A- greater than, B-less than C. A- less than, B- greater than D. A- less than, B- less than E.
HCN Lewis Structure, Molecular Geometry, Shape, and Polarity Carbon forms one single bond with the Hydrogen atom and forms a triple bond with the Nitrogen atom. · HCN has a total of 10 valence electrons.Jul 23, 2021 · Uploaded by Geometry of Molecules www2.chemistry.msu.edu › faculty › reuschAlkyl Halide Reactivity - Michigan State University An energy diagram for the single-step bimolecular E2 mechanism is shown on the right. We should be aware that the E2 transition state is less well defined than is that of S N 2 reactions. More bonds are being broken and formed, with the possibility of a continuum of states in which the extent of C–H and C–X bond-breaking and C=C bond-making ...


























0 Response to "39 lewis diagram for hcn"
Post a Comment